Field-Flow Fractionation (FFF) is an elegant separation technique because it is a simple concept that is extremely broad in practice:
An external transverse force is applied across a channel to a solution of solutes (maybe colloids, maybe polysaccharides maybe cells, whatever). The perpendicular field pushes the ensemble of solutes against the bottom wall but diffusion disperses the solutes and resists the inhomogeneity. The competition between thermal and potential energy results in an exponential concentration distribution in equilibrium. Importantly, each species of solute has a different concentration profile (perhaps because they have different masses if gravity or centerfugation is being used or because they have different charge if an electric field is being used, etc).
If nonuniform flow profile carries these solutes through the channel, their elution times will differ. Since each species has a different concentration distribution, each species samples the velocity distribution with a different weighting and so has a different mean velocity. Solute particles near the wall are subject to slower flow than particles near the centre; therefore, samples with a mean height close to the wall are carried along with a slower average velocity than samples with a larger mean height.
This seems simple enough but interestingly this simple system possesses surprisingly rich elution behaviour.
Hydrodynamic Chromatography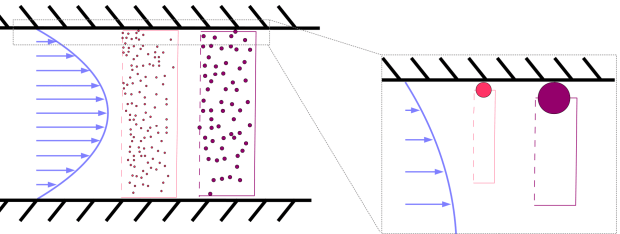
Imagine that the external field is turned off or negligible. In this case the solutes can diffuse across the entire extent of the channel and uniformly sample the parabolic velocity distribution. EXCEPT that there is an excluded region near the walls. If the solutes are hard spheres then there is a stericly excluded film that is exactly equal to their radii. This excluded zone changes with particle size and so the average solute velocity is different. This is the basis of Hydrodynamic Chromatography.
Normal-Mode FFF
 If instead we imagine that the field is strong and the particles are small (so small that we might model them as point particles). Then we only need to average the parabolic fluid velocity weighted by an exponential probability distribution. The particle velocity wouldn’t have an explicit size dependence (since it would be modelled as a point particle) but only a external force dependence.
If instead we imagine that the field is strong and the particles are small (so small that we might model them as point particles). Then we only need to average the parabolic fluid velocity weighted by an exponential probability distribution. The particle velocity wouldn’t have an explicit size dependence (since it would be modelled as a point particle) but only a external force dependence.
Steric-Mode FFF
 As the particles get larger and larger, the point-particle approximation breaks down and one needs to include both the external force on the colloids and the steric force when they interact with the wall.
As the particles get larger and larger, the point-particle approximation breaks down and one needs to include both the external force on the colloids and the steric force when they interact with the wall.
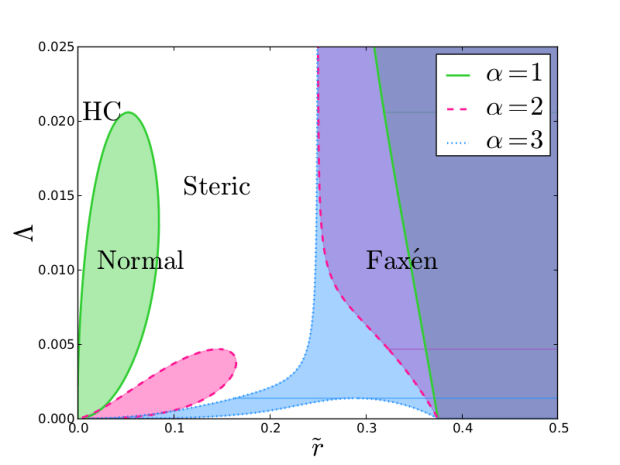
We were able to combine all three of these “operational-modes” of FFF into a single unified theory by simply writing the retention theory explicitly in terms of particle size. When this is done FFF retention theory can predict all three modes and the transitions between these modes.
Faxén-Mode FFF
 We were happy to have a unified theory but found that when we let the particle size approach the channel height the particle was predicted to go 50% faster than the fluid — a highly unlikely thing. So we went back and added a bit more hydrodynamics: We integrated the stress on the particle due to the fluid flow. Because the fluid profile is parabolic, the particle moves slightly slower than the carrier fluid, which of course means that the particles elute later than if this effect were neglected.
We were happy to have a unified theory but found that when we let the particle size approach the channel height the particle was predicted to go 50% faster than the fluid — a highly unlikely thing. So we went back and added a bit more hydrodynamics: We integrated the stress on the particle due to the fluid flow. Because the fluid profile is parabolic, the particle moves slightly slower than the carrier fluid, which of course means that the particles elute later than if this effect were neglected.
We mapped out the transitions between these ideal modes of operation.
Find more details or the citation for this in our article in the Journal of Chromatography A.

2 thoughts on “Unified, Ideal FFF”
Comments are closed.